How to Become a Clinical Research Coordinator with no Experience

Learn how to become a Clinical Research Coordinator with no experience through training, CRC certification, skills, and career guidance.
Clinical research is creating strong career opportunities for freshers, healthcare graduates, and professionals looking to enter the pharmaceutical and clinical trials industry. Among the most in-demand roles in this field is the Clinical Research Coordinator (CRC).
Many candidates believe that entering this profession requires years of hospital or research experience. In reality, structured training, practical understanding of clinical trials, and industry-relevant skills can help beginners start their journey successfully.
If you want to become a clinical research coordinator without experience, this guide explains the exact roadmap, qualifications, skills, certifications, and career steps needed to enter the field confidently.
What Does a Clinical Research Coordinator Do?
A Clinical Research Coordinator manages and supports clinical trial activities at hospitals, research sites, or healthcare institutions. They act as a bridge between investigators, sponsors, patients, and ethics committees to ensure studies are conducted according to protocol and regulatory guidelines.
Their responsibilities usually include:
- Coordinating clinical trial activities
- Screening and scheduling patients
- Maintaining trial documentation
- Managing informed consent forms
- Ensuring protocol compliance
- Handling data collection and reporting
- Communicating with sponsors and CROs
- Supporting site audits and monitoring visits
Clinical Research Coordinators play a major role in maintaining study quality, patient safety, and regulatory compliance throughout the clinical trial process.
Can You Become a Clinical Research Coordinator Without Experience?
Yes, it is possible to become a Clinical Research Coordinator without prior experience.
Many organizations hire freshers who possess:
- Strong understanding of clinical research fundamentals
- Knowledge of GCP and clinical trial processes
- Good communication and documentation skills
- Professional certification or structured CRC training
- Basic understanding of medical terminology and regulations
The clinical research industry values practical knowledge and trained candidates who can adapt quickly to trial environments. This is why certification-focused learning programs have become increasingly important for freshers entering the industry.
Educational Qualification Required for CRC Roles
For India:
Common educational qualifications include:
- Life Sciences (B.Sc., M.Sc.)
- Pharmacy (B.Pharm, M.Pharm)
- Nursing or Allied Healthcare
- Biotechnology or Microbiology
- BDS, BHMS, BAMS (in some cases)
These backgrounds provide foundational knowledge in biology, patient care, and medical terminology, which are useful in clinical research.
For USA:
Typical educational requirements include:
- Bachelor’s degree in Life Sciences (Biology, Chemistry, etc.)
- Nursing (BSN or equivalent)
- Public Health or Health Sciences
- Pharmacy or related healthcare fields
In many cases, employers in the USA prefer candidates with:
- A bachelor’s degree as a minimum qualification
- Additional certification or clinical research training
- Some level of healthcare or research exposure
Important Note: Candidates from non-science backgrounds can also enter clinical research through structured training and CRC certification programs, making it a viable option for career switchers. A strong educational foundation, combined with practical understanding, plays a key role in becoming a successful Clinical Research Coordinator.
Essential Skills Needed to Become a Clinical Research Coordinator
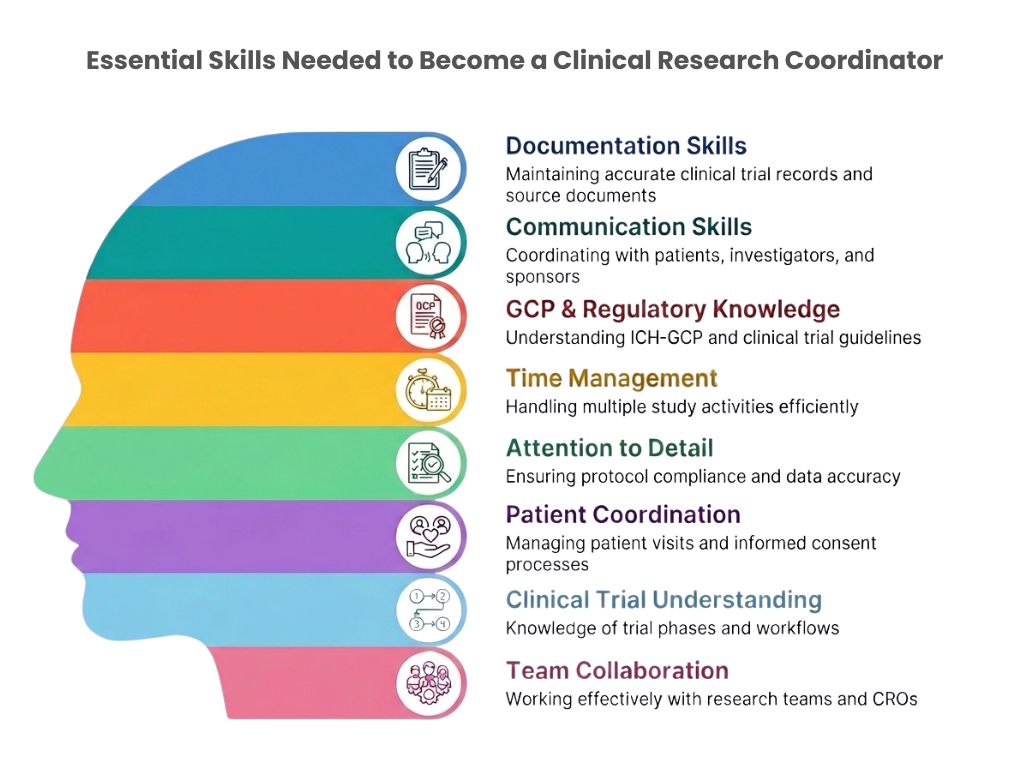
Even without experience, developing the right skills can significantly improve employability.
Communication Skills
CRC professionals regularly interact with patients, investigators, sponsors, and ethics committees. Clear and professional communication is essential.
Documentation and Organizational Skills
Clinical trials involve extensive documentation. Maintaining accurate records and handling study files properly is one of the core responsibilities of a CRC.
Attention to Detail
Minor documentation or protocol errors can impact study quality. Employers prefer candidates who demonstrate accuracy and responsibility.
Understanding of Clinical Trial Processes
Candidates should understand:
- Clinical trial phases
- Informed consent process
- Adverse event reporting
- ICH-GCP guidelines
- Protocol compliance
- Source documentation
Time Management Clinical Research Coordinators often manage multiple activities simultaneously, including patient visits, documentation, sponsor communication, and study timelines.
Step-by-Step Guide to Becoming a Clinical Research Coordinator
Entering the clinical research industry as a fresher becomes easier with the right guidance, training, and practical understanding. The following roadmap explains the essential steps to start a career as a Clinical Research Coordinator, even without prior experience.
| Step | What You Need to Do | Why It Matters |
| Step 1 | Learn clinical research fundamentals | Builds foundational knowledge |
| Step 2 | Join a CRC certification program | Improves practical understanding |
| Step 3 | Gain internship or practical exposure | Helps build industry confidence |
| Step 4 | Prepare a professional resume | Improves job opportunities |
| Step 5 | Apply for entry-level CRC roles | Starts your clinical research career |
Step 1: Build Strong Clinical Research Fundamentals
The first step is understanding how clinical trials work. Beginners should focus on learning:
- Drug development process
- Clinical trial phases
- Roles in clinical research
- Ethics and patient safety
- Regulatory guidelines
- Clinical trial documentation
A strong foundation helps candidates perform confidently during interviews and internships.
Step 2: Join a Structured CRC Certification Program
One of the most effective ways to enter the industry without experience is through a specialized Clinical Research Coordinator certification program.
A professional training course helps candidates gain:
- Industry-oriented knowledge
- Understanding of real clinical trial workflows
- Exposure to documentation practices
- GCP and regulatory understanding
- Confidence for interviews and job applications
Today, many learning platforms offer Clinical Research Coordinator certification programs online in a self-paced and affordable format for freshers and aspiring clinical research professionals. One such example is the Clinical Research Coordinator Certification offered by ProRelix Education, which is designed to provide practical, job-oriented learning aligned with current clinical research industry expectations.Such structured certification programs help bridge the gap between academic education and real-world clinical research requirements.
Here is your Clinical Research Coordinator Certification sample, designed to support practical learning and career readiness in clinical research.

Download Clinical Research Coordination Certifiaction Training Syllabus
What Should a Good CRC Certification Course Include?
Before selecting any training program, candidates should look for:
- Clinical trial fundamentals
- ICH-GCP training
- Regulatory guidelines
- Source documentation practices
- Case-study-based learning
- Site coordination workflows
- Patient recruitment concepts
- Adverse event handling
- Industry-oriented practical exposure
Programs focused on real clinical operations are generally more valuable for entry-level candidates.
Step 3: Gain Practical Exposure
Even if you do not have formal experience, you can still gain industry exposure through:
- Internships
- Site-based observation programs
- Clinical research workshops
- Documentation practice sessions
- Mock case studies
- Research projects
Practical understanding improves confidence and strengthens your resume during job applications.
Step 4: Prepare an Industry-Focused Resume
Freshers often underestimate the importance of a professional clinical research resume.
Your resume should highlight:
- Clinical research certification
- GCP training
- Academic projects
- Internship exposure
- Communication skills
- Documentation knowledge
- Healthcare or patient-handling experience
A well-structured resume can improve shortlisting chances even without prior industry experience.
Step 5: Apply for Entry-Level Clinical Research Roles
After training, candidates can apply for:
- Clinical Research Coordinator trainee roles
- Clinical Trial Assistant positions
- Site Coordinator roles
- Clinical Research Internships
- Clinical Documentation support roles
- Research assistant positions
Hospitals, Contract Research Organization(CROs), SMOs, and research institutes regularly hire trained freshers for entry-level opportunities.
How to Prepare for CRC Interviews Without Experience
Interviewers usually assess:
- Understanding of clinical trial basics
- Knowledge of GCP
- Communication ability
- Professional attitude
- Interest in clinical research
- Understanding of CRC responsibilities
Candidates should prepare answers for questions such as:
- What is informed consent?
- What are adverse events?
- What is the role of a CRC?
- Why do you want to enter clinical research?
- What is ICH-GCP?
Practical understanding matters more than memorized definitions.
Salary and Career Growth for Clinical Research Coordinators
CRC is considered one of the strongest entry points into the clinical research industry, especially for freshers entering the healthcare and pharmaceutical sectors.
| Experience Level | India Salary Range | USA Salary Range | Career Growth Opportunities |
| Entry-Level CRC | ₹2.5 LPA – ₹4.5 LPA | $45,000 – $65,000 | CRC Trainee, Clinical Trial Assistant, Site Coordinator |
| Mid-Level CRC | ₹6 LPA – ₹12 LPA+ | $75,000 – $120,000+ | Senior CRC, CRA, Project Coordinator, Site Manager |
| Advanced Clinical Operations Roles | ₹12 LPA+ | $120,000+ | Clinical Trial Manager, Clinical Operations Specialist, Project Manager |
Entry-Level Salary
The salary of a Clinical Research Coordinator depends on factors such as educational background, location, organization type, certification, and practical knowledge.
In India
Freshers entering CRC roles in India typically earn between ₹2.5 LPA to ₹4.5 LPA in entry-level positions. Candidates with structured clinical research training, internship exposure, or CRC certification may have better opportunities for higher starting packages.
In the USA
Entry-level Clinical Research Coordinators in the United States generally earn between $45,000 to $65,000 per year, depending on the state, healthcare institution, research organization, and prior training exposure.
Career Growth Opportunities
With experience, CRC professionals can progress into roles such as:
- Senior Clinical Research Coordinator
- Clinical Trial Manager
- Clinical Research Associate (CRA)
- Project Coordinator
- Site Manager
- Clinical Operations Specialist
Experienced professionals may earn between ₹6 LPA to ₹12 LPA+ in India and $75,000 to $120,000+ annually in the USA, depending on specialization, therapeutic area, and organization type.Professionals with strong documentation skills, GCP understanding, clinical trial workflow knowledge, and relevant certification often receive better career opportunities and faster salary growth in hospitals, CROs, research sites, universities, and multinational clinical research organizations.
Why Clinical Research Certification Is Valuable for Freshers
Certification adds credibility to a candidate’s profile and demonstrates commitment toward the profession.
For beginners, certification helps:
- Build industry confidence
- Understand practical workflows
- Improve interview readiness
- Strengthen resumes
- Increase employability
- Gain structured clinical research knowledge
This is especially important for candidates entering the field without direct experience.
Final Thoughts
Starting a career as a Clinical Research Coordinator without experience is possible with the right learning approach, industry understanding, and practical preparation.
The clinical research industry values candidates who possess strong fundamentals, professional communication skills, regulatory awareness, and willingness to learn. By combining structured training with practical exposure, freshers can successfully enter this growing field and build long-term careers in clinical research.For aspiring professionals looking for structured industry-oriented training, the Certification for CRC program by ProRelix Education can serve as a strong starting point toward understanding real clinical research workflows and preparing for entry-level opportunities in clinical trials.
FAQs
Yes, you can become a Clinical Research Coordinator with no experience by gaining clinical research knowledge, CRC certification, and practical understanding of clinical trial processes.
To become a Clinical Research Coordinator, candidates usually need backgrounds in pharmacy, life sciences, biotechnology, nursing, or other healthcare-related fields.
Yes, CRC certification is important for freshers because it helps build clinical research knowledge, GCP understanding, documentation skills, and interview confidence.
The salary of a Clinical Research Coordinator in India typically starts from ₹2.5 LPA to ₹4.5 LPA, while in the USA it may range from $45,000 to $65,000 annually.
The time required to become a Clinical Research Coordinator depends on training and practical learning, but many candidates can start applying for entry-level CRC roles within a few months after certification.


