Clinical Research Courses in Pune with Placements at IQVIA, TCS, Cognizant, and More
ProRelix Education provides the best Clinical Research Courses in Pune with Placements with the objective of preparing individuals for the pharmaceutical and healthcare work environments. The strong focus on practical training and real industry exposure ensures that graduates have high chances of securing jobs in leading organizations like IQVIA, TCS, Cognizant and many more.
Numerous students have already availed these opportunities and achieved their dream careers. Many proudly share success stories of how ProRelix’s complete training and placement support became the key reason for their growth and confidence in the clinical research industry.
Get Free Consultation
ProRelix Education is an award winning institute offering Best Clinical Research Courses In Pune with Placements opportunities at top tier MNCs and CROs. We provide both online and offline learning programs focused on practical training, industry exposure and global clinical research standards.
Our training approach is strengthened by recognized accreditations and industry partnerships. ProRelix Education signed an MoU with the Life Sciences Sector Skill Development Council (LSSSDC) in 2017 and is ISO 9001:2015 certified. The institute was awarded “Best Institute for Clinical Research” at the Asia Education Summit & Awards and also received a “Certificate of Commitment” from the Central Vigilance Commission of India in 2016. We have also signed agreements with multiple organizations in the UK, South Africa, USA and Peru to support clinical research development initiatives, enhancing global exposure for learners.
With a strong focus on ethical practices, career readiness and continuous learning, ProRelix Education supports students and working professionals in building successful careers in the clinical research and healthcare industry.
Clinical Research Course Highlights
- Top Quality at the Best Price
- 100% Placement Assistance
- Unlimited Job Opportunity Calls
- Learn Directly from Industry Professionals
- Proven 90% Placement Success Rate
- Flexible Payment Plans Available
- 10+ Years of Industry Experience
- Own ProRelix Research CRO for Internship Opportunities
- Development in Interview, CV, and Presentation Skills
Offline Fulltime Clinical Research Course in Pune with Placement Assistant
Modes of Teaching
Offline Classroom
Course Duration
04 Months
Courses Fees
40K -60K
Placement
100% Assistance
Student Trained
10000+
Online Fulltime Clinical Research Course in Pune with Placement Assistant
Modes of Teaching
Online Live
Course Duration
04 Months
Courses Fees
40K -60K
Placement
100% Assistance
Student Trained
10000+
ProRelix Education offers a full-time PG Diploma in Clinical Research Course in Pune with placements opportunities at leading companies such as IQVIA, Lybra Clinical Research, Tata Consultancy Services, Wellbeing Research Centre, Raptim Research, ProRelix Research, Parexel, IKS Health, Sahyadri Hospitals Cognizant and more. This program, also known as the PGDCR: Post Graduate Diploma In Clinical Research, has a duration of 4 months and is available through both online live classes and offline classroom sessions in Karvenagar, Pune with placement opportunitis. The course fees range from 40,000 to 50,000 INR, depending on the selected modules and internship program. Many students have benefited from this course and its internship opportunities, securing placements in top companies.
PGDCR Clinical Research Course Syllabus [2025 Edition]
PGDCR Clinical research course content offered by ProRelix Education is structured to align with current regulatory frameworks and global clinical trial standards, with a strong focus on conceptual clarity, practical understanding, and job-oriented skills. The syllabus is designed to prepare learners for roles across clinical trials, data management, and pharmacovigilance.
1 : Fundamentals of Clinical Research
- Introduction to Clinical Research
- History & evolution of Clinical Research
- Drug development process and preclinical studie
- Phases of clinical trials and BA/BE studies
- Stakeholders in clinical research and their roles (Sponsor, Investigator, CRO, Ethics committees etc.
- Types of clinical trials and basic trial designs
- Regulatory framework and clinical trial registries
- Ethics in clinical research and informed consent
- Essential documents (Protocol, ICF, CRF, IB, etc.)
- ICH GCP E6 R
- Good Documentation Practices and ALCOA
- Basics of safety reporting
- Terminologies related to Clinical Research
2 : Clinical Research Operations
- Site selection & feasibility
- Initiation and ethics approvals
- Subject recruitment and clinical trial conduct
- Monitoring activities and source data verificatio
- Types of monitoring
- Investigational product management and accountabilit
- Protocol deviations and safety reporting
- Quality management, audits, and inspections
- Trial close-out, documentation, and archiving
3 : Clinical Data Management
- Introduction to Clinical Data Management & its role in clinical trials
- Clinical data management process flow and data lifecycle
- Data collection methods and instruments
- Data management team roles and responsibilities
- Data Management Plan (DMP)
- CRF/eCRF design principles and CDASH standards
- Edit check specifications and validation rules
- EDC systems, database build, and User Acceptance Testing (UAT)
- Data entry, validation, and query management
- Medical coding and safety data reconciliation
- Computer System Validation (CSV) and 21 CFR Part 11
- Database lock, data transfer, and study close-out activities
- CDM quality management, documentation, and compliance
4 : Pharmacovigilance
- Introduction to Pharmacovigilance
- Adverse events, special situations, and case validity
- Causality & seriousness assessment
- Individual Case Safety Reports (ICSRs)
- Case Processing and Coding
- Safety Reporting & timelines
- Signal Detection and Risk Management
- Pharmacovigilance Systems and Quality Management
- Pharmacovigilance regulations in India and global context
- Pharmacovigilance Databases and Tools
- Common terminologies related to pharmacovigilance
5 : Regulatory Affairs
- Introduction to Pharmacovigilance
- Adverse events, special situations, and case validity
- Causality & seriousness assessment
- Individual Case Safety Reports (ICSRs)
- Case Processing and Coding
- Safety Reporting & timelines
- Signal Detection and Risk Management
- Pharmacovigilance Systems and Quality Management
- Pharmacovigilance regulations in India and global context
- Pharmacovigilance Databases and Tools
- Common terminologies related to pharmacovigilance
Our Placed Students
Our students have placements with leading companies worldwide.









Top Career Opportunities After Clinical Research Course Done
1 : Clinical Trial Associate
A Clinical Trial Associate supports the planning, coordination, and documentation of clinical trials to ensure compliance with regulatory guidelines, protocols, and timelines.
2 : Clinical Research Coordinator
A Clinical Research Coordinator (CRC) manages and supports the day-to-day operations of clinical trials, ensuring protocol compliance, accurate data collection, and participant safety.
3 : Clinical Research Associate
A Clinical Research Associate (CRA) is responsible for monitoring clinical trials to ensure they are conducted ethically, accurately, and in compliance with regulatory guidelines and study protocols.
4 : Data Safety Associate
A Data Safety Associate supports clinical trials by monitoring, reviewing, and ensuring the accuracy, compliance, and integrity of clinical safety data in line with regulatory and quality standards.
5 : Regulatory Associate
A Regulatory Associate supports regulatory submissions, ensures compliance with national and international guidelines, and coordinates with regulatory authorities throughout the product lifecycle.
6 : Investigators
Investigators are qualified medical professionals responsible for conducting clinical trials at study sites, ensuring participant safety, protocol compliance, and accurate data collection throughout the study.
7 : Medical Writer
A Medical Writer develops clear, accurate scientific content for clinical research, regulatory documents, and healthcare communications.
8 : Business Development Executive
A Business Development Executive is responsible for identifying new business opportunities, building client relationships, and driving company growth through strategic sales and partnerships.
9 : Quality Assurance Associate
A Quality Assurance Associate ensures compliance with regulatory standards by monitoring processes, reviewing documentation, and maintaining quality systems to support accurate and ethical clinical research activities.
10 : Clinical Data Associate
A Clinical Data Associate is responsible for collecting, validating, and managing clinical trial data to ensure accuracy, consistency, and compliance with regulatory standards throughout the study lifecycle.
Clinical Research Course Program Learning at ProRelix Education
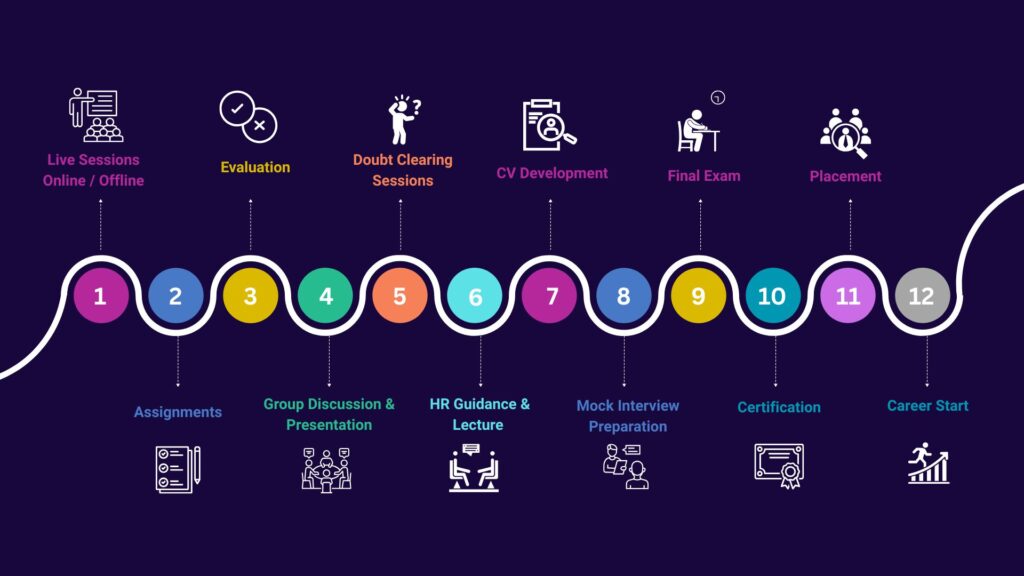
Our Placement Companies














Frequently Ask Questions (FAQ's)
What are clinical research courses?
Clinical research courses provide knowledge and training in conducting and managing clinical trials for new drugs, therapies, and medical devices, ensuring safety and efficacy.
Who can enroll in clinical research courses?
Graduates in life sciences, pharmacy, biotechnology, medicine, or related fields are eligible to enroll in clinical research courses.
What topics are covered in clinical research courses?
Topics include drug development, clinical trial phases, regulatory guidelines (ICH-GCP), pharmacovigilance, and data management.
Where can I find clinical research courses in Pune?
Institutes like ProRelix Education in Pune offer comprehensive clinical research courses with certification and placement support.
Are there classroom and online clinical research courses in Pune?
Yes, ProRelix Education institute in Pune provide both classroom-based and online options for clinical research courses to suit different needs.
What is the duration of clinical research courses in Pune?
The duration varies from short-term certification courses (1-3 months) to PG diplomas (6-12 months).
What are the benefits of online clinical research courses?
Flexibility, accessibility, and opportunities to learn from global experts are key benefits of online clinical research courses.
Can I start a career in clinical research with free courses?
Free courses can be a good starting point, but advanced certifications or diplomas are typically needed for a clinical research career.
Do clinical research courses include placement assistance?
Yes, many institutes, including ProRelix Education, offer placement support after course completion.
Students Feedback
Trainee Clinical Data Coordinator (IQVIA)
I’ve completed my course at ProRelix Education. It was a wholesome experience of learning. The faculty is helpful and teaches very well, the trainers are available all time for query resolution.
Pharmacovigilance Trainee (TCS)
End-to-end query resolution, and placement assistance from the beginning. They also conduct group discussions, and quiz competitions to improve communication skills of students. Thanks for the opportunity! for query resolution.
Junior Data Analyst (IKS Health)
The courses are excellent and easy to understand; the trainers are knowledgeable and helpful; would recommend anyone learning about deep clinical research knowledge. of students. Thanks for the opportunity! for query resolution.
