
The clinical research industry is expanding rapidly across the globe, creating exciting opportunities for professionals with the right educational background and technical expertise. Pursuing an Advanced Diploma in Clinical Research is one of the best ways to enter this growing sector and build a successful international career in clinical trials.
ProRelix Education offers an Advanced Diploma in Clinical Research to make you job ready with global certification, expert mentors, and assured placement support.
If you are a pharmacy, life sciences, nursing, or medical graduate aiming to build a career in drug development, clinical operations, or pharmacovigilance, this course can be your gateway to the global healthcare industry.
Why Choose an Advanced Diploma in Clinical Research?
An Advanced Diploma in Clinical Research equips students with advanced knowledge of clinical trial design, global regulations, and practical skills, preparing them for high-level roles in pharmaceutical companies, CROs, and research organizations worldwide. The Post Graduate Diploma in Clinical Research, one of the best Advanced Diploma programs offered by ProRelix Education, provides in-depth learning on how clinical trials are designed, conducted, and managed according to international regulatory guidelines, helping students gain both theoretical and practical expertise for a successful global career.
Key Learning Outcomes:
- Thorough understanding of GCP (Good Clinical Practice) and regulatory guidelines
- Expertise in clinical trial management and monitoring
- Knowledge of pharmacovigilance, data management, and biostatistics
- Insights into drug safety and regulatory submissions
- Understanding of ethical considerations and patient safety in research
- Real-world exposure through ProRelix Research projects, offering practical experience in ongoing clinical studies
- Global certification accepted in the USA, UK, and Europe, adding international credibility to your qualification
This program not only enhances your job readiness but also helps you qualify for roles in pharmaceutical companies, contract research organizations (CROs), hospitals, and regulatory agencies.
What ProRelix Education Offers in the Advanced Diploma in Clinical Research
ProRelix Education offers the Post Graduate Diploma in Clinical Research (PGDCR) as its Advanced Diploma in Clinical Research, designed to provide both theoretical knowledge and hands-on training for aspiring clinical research professionals. The program combines the essential elements of clinical research, data management, and pharmacovigilance, helping students build the skills required for high-level roles in the pharmaceutical and healthcare industries.
Program Overview
- Full-time and Part-time Options: Flexible learning with internship opportunities.
- Classroom Training: Delivered through high-quality presentations by experienced industry experts.
- Objective: A 6-month, in-depth, industry-oriented program to prepare students for leadership roles in the clinical research field.
- Curriculum: Integrates Clinical Research, Clinical Data Management, and Pharmacovigilance, along with Interview Techniques and CV Development Workshops to ensure a successful career path.
- Industry-Aligned Structure: Designed to meet global academic and industry standards.
Teaching Methodology
- Classroom Lectures
- Presentations and Assignments
- Project Work
- Language Options: English, Hindi, Marathi
- 24/7 Assistance via Skype
- Limited Seats for Personalized Attention
- CV and Personal Development Sessions
Program Duration
- Total Duration: 6 Months
- 4 Months: Intensive Training
- 2 Months: Internship
- 4 Months: Intensive Training
- Schedule: Monday to Thursday
- First Batch: 10:30 AM – 12:30 PM
- Second Batch: 2:00 PM – 4:00 PM
- First Batch: 10:30 AM – 12:30 PM
Program Benefits
- Well-equipped Training Center
- Academic and Personal Growth
- Free Study Materials
- Internship Opportunities
- Placement Assistance
- Internationally Recognized Certificate
PGDCR Program Syllabus
An industry-centric syllabus ensures students gain practical knowledge and exposure aligned with the latest trends and requirements in clinical research.
Eligibility
Graduates from pharmacy, life sciences, medicine, biotechnology, and related fields are eligible to enroll in this program.
Placement Support
ProRelix Education provides comprehensive placement support to help students connect with leading pharmaceutical companies, CROs, and hospitals, ensuring a strong start to their clinical research careers.
Who Can Enroll in the Advanced Diploma Program?
This course is ideal for:
- B.Pharm, M.Pharm, B.Sc., M.Sc., MBBS, BDS, BAMS, BHMS, or life science graduates
- Fresh graduates looking for an entry point into clinical research
- Working professionals seeking to upskill and shift to clinical trial management
Whether you’re a beginner or already working in the healthcare domain, this diploma helps you gain global exposure and professional recognition in the clinical research field.
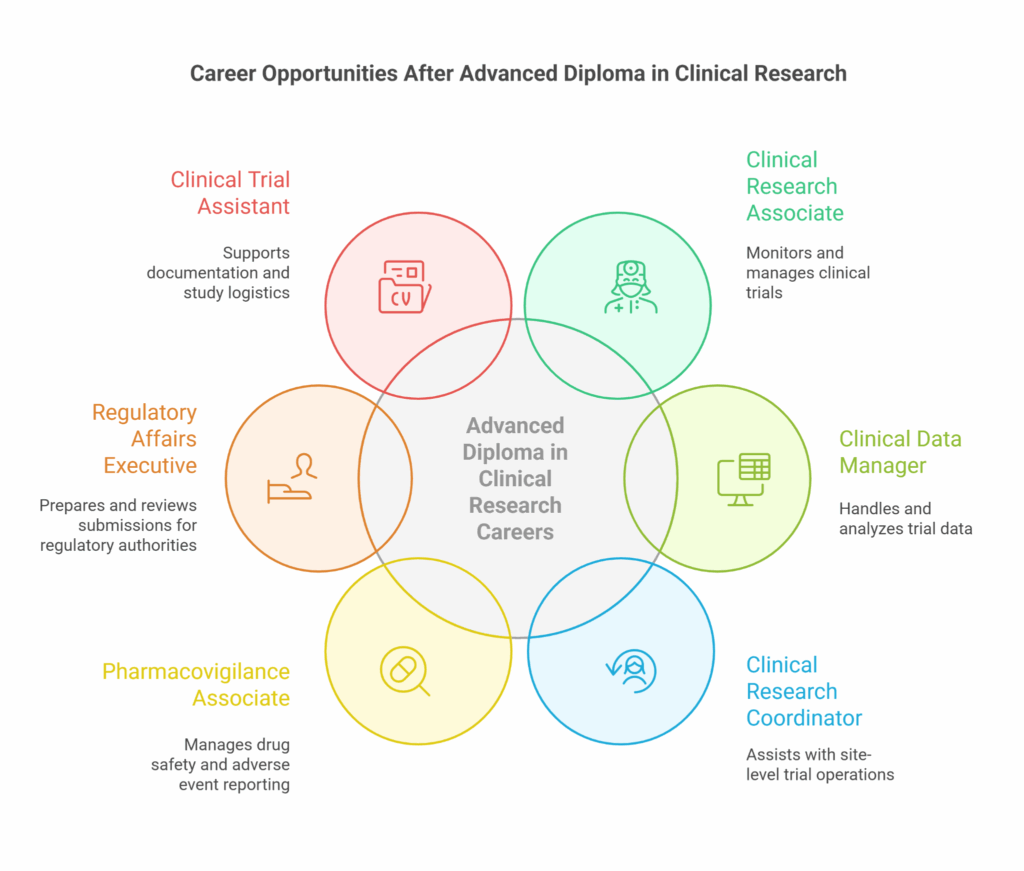
Career Opportunities After Advanced Diploma in Clinical Research
After completing this program, you can pursue several promising roles such as:
| Job Role | Primary Responsibilities |
| Clinical Research Associate (CRA) | Monitoring and managing clinical trials |
| Clinical Data Manager | Handling and analyzing trial data |
| Clinical Research Coordinator | Assisting with site-level trial operations |
| Pharmacovigilance Associate | Managing drug safety and adverse event reporting |
| Regulatory Affairs Executive | Preparing and reviewing submissions for regulatory authorities |
| Clinical Trial Assistant | Supporting documentation and study logistics |
With growing investments in global healthcare and research, the demand for trained clinical research professionals continues to increase every year especially in India, the USA, Europe, and Asia-Pacific regions.
Demanding Self-Paced Learning Programs by ProRelix Education
If you are just beginning your journey or looking to advance your skills in the field of clinical research, ProRelix Education offers a range of self-paced and structured online programs designed to support your professional growth. Below are some of the most demanding self-paced learning programs offered by ProRelix Education.
The Beginner Course in Clinical Research is the ideal starting point for students who want to understand the fundamentals of clinical trials, research methodologies, and regulatory processes. Each program is designed to provide flexible learning, industry-relevant knowledge, and globally recognized certification helping students and professionals gain the expertise required to build successful careers in clinical research and data management.
Beginner Course in Clinical Research (Certification Program):
Highlights:
- 100% online learning with flexible access
- Basics of clinical trials, ICH-GCP, and ethics
- Designed for freshers from life sciences, pharmacy, and medical backgrounds
- Certification upon completion
This course helps you step confidently into the clinical research industry and prepares you for entry-level roles like Clinical Research Coordinator or Clinical Trial Assistant.
Explore Beginner Course in Clinical Research
Advanced Course in Clinical Research (Professional Certification)
Once you’ve mastered the basics, the Advanced Course in Clinical Research helps you develop deeper expertise in clinical trial monitoring, site management, and regulatory documentation.
What You’ll Learn:
- Advanced GCP principles and trial operations
- Data interpretation and reporting
- Real-world exposure through ProRelix Research projects
- Global certification accepted in the USA, UK, and Europe
Perfect for working professionals and those aiming for roles like Clinical Research Associate (CRA) or Regulatory Affairs Executive.
Explore Advanced Course in Clinical Research
ICH-GCP Good Clinical Practice Online Certification
Good Clinical Practice (GCP) is the backbone of every successful clinical trial. The ICH-GCP Online Certification Course from ProRelix Education ensures that you understand the ethical and scientific quality standards required for conducting global trials.
Course Benefits:
- Learn global ICH-GCP guidelines
- Understand investigator and sponsor responsibilities
- Compliant with WHO and regulatory standards
- Short-term certification recognized internationally
This program is essential for anyone planning to work in CROs, hospitals, or pharmaceutical companies involved in research.
Beginner Course in Clinical Data Management (CDM)
The Beginner Course in Clinical Data Management introduces you to the world of managing, validating, and analyzing clinical trial data.
Program Overview:
- Learn about CRFs, data validation, and query management
- Understand EDC tools and data flow in trials
- Practical assignments to build real skills
- Industry-recognized certification
With the growing need for data professionals, this course prepares you for a career as a Clinical Data Coordinator or Data Manager.
Explore Beginner Course in CDM
Why Choose ProRelix Education for Clinical Research Courses?
ProRelix Education is one of India’s leading and trusted institutes offering specialized courses in clinical research, pharmacovigilance, clinical data management, and regulatory affairs. With over a decade of experience, the institute is known for providing globally recognized certification programs and placement assistance with top CROs and pharmaceutical companies. ProRelix Education also offers clinical research online courses, including the Beginner Course in Clinical Research (Certification Program), designed for students who prefer flexible and remote learning options.
Highlights of ProRelix Education’s Clinical Research Programs:
- Industry-aligned curriculum designed by experienced professionals
- Online and offline learning modes for flexible study
- Hands-on practical training and case study-based learning
- Expert faculty from clinical and pharmaceutical domains
- Internship opportunities and placement support
Students from ProRelix Education have successfully built global careers in India, the Middle East, Europe, and North America, making it a preferred choice for aspiring clinical research professionals.
India – A Global Destination for Clinical Research Education
India has become a major hub for clinical research and trials due to its large patient pool, skilled workforce, and cost-effective operations. As pharmaceutical companies continue to expand their R&D operations, the need for qualified clinical research professionals is increasing rapidly.
Institutes like ProRelix Education bridge this skill gap by providing job-oriented programs aligned with international standards, enabling Indian students to compete and succeed globally.
Final Thoughts
The Post Graduate Diploma in Clinical Research (PGDCR), offered as an Advanced Diploma in Clinical Research by ProRelix Education, is a job-oriented program designed to build global careers in clinical research, pharmacovigilance, and regulatory affairs. Through expert mentorship, practical training, and assured placement support, the program equips students with the essential skills, international certification, and industry exposure required to excel in the rapidly growing clinical research sector.
Start Learning with ProRelix Education Today
Take your next step in the world of clinical trials and global drug research.
Enroll in a Post Graduate Diploma in Clinical Research (PGDCR) full-time course with ProRelix Education and gain in-depth industry knowledge, live mentorship from experts, and dedicated career support to build a successful career in the clinical research industry.
Contact: +91 7304 555 555
Email: training@prorelixeducation.com
Website: www.prorelixeducation.com


