Online Clinical Data Management Course
The Online Clinical Data Management Course is designed to students and professionals with in-depth knowledge of managing clinical trial data as per global regulatory standards. The course covers key areas such as CDM processes, CRF design, data validation, database management, medical coding, and regulatory compliance (ICH-GCP, FDA, CDISC). ProRelix Education ensures career focused skill development along with placement guidance to help learners succeed in the clinical research industry.
Certified Online Clinical Data Management Course with Industry Exposure
ProRelix Education offers a Certified Online Clinical Data Management Course with Industry Exposure to help learners build job-ready skills for the evolving clinical research industry. It covers core concepts such as clinical data lifecycle, CRF design, data validation, database management, CDISC standards, and regulatory compliance, ensuring learners gain strong theoretical and practical knowledge. Delivered through expert-led online sessions, the course offers flexibility while maintaining a strong focus on real-world applications.
This online CDM course is its industry exposure, including practical assignments, real-time case studies, and hands-on training aligned with current industry practices.This program emphasizes job readiness by bridging the gap between academic learning and industry expectations, helping learners build confidence and practical expertise. This clinical data management certification is ideal for graduates, life science professionals, and working individuals seeking career growth in clinical data management roles within CROs, pharmaceutical companies, and healthcare organizations.
Key Features
- Flexible online learning for self-paced study and convenience
- Learn updated clinical data management processes and regulatory compliance
- Programs developed by professionals with 20+ years of industry experience
- Certificate of completion to boost career opportunities
- Modules covering SDTM, CDASH, and data validation
- Online support
How to Enroll in Online Clinical Data Management Course
Enrolling in the Online Clinical Data Management Course at ProRelix Education is a simple and seamless process designed to get you started on your career in clinical research quickly. Our course is crafted for working professionals, students, and fresh graduates who want flexible learning with real-world industry exposure.
Step 1: Visit the Course Page
Go to the Online Clinical Data Management Course page on the ProRelix Education website. Here, you can explore the course curriculum, key features, and benefits of learning with industry experts.
Step 2: Fill the Registration Form
Click on the “Get Course” button and fill out the registration form with your details. Make sure to provide accurate information to complete the enrollment successfully.
Step 3: Make Payment
Click on the “Get Course” button and fill out the registration form with your details. Make sure to provide accurate information to complete the enrollment successfully.
Step 4: Confirmation and Course Access
Once your payment is confirmed. You can immediately access the course modules, study materials, and assignments to begin your journey in Clinical Data Management.
Advanced Online Clinical Data Management Course with Certification
Elevate your career in clinical research with our Advanced Online Clinical Data Management Course, designed to provide comprehensive knowledge and hands-on experience in managing clinical trial data efficiently. This course is ideal for professionals aiming to gain expertise in clinical data management (CDM) and secure a strong foothold in the pharmaceutical and healthcare industries.
Our curriculum covers advanced topics including electronic data capture (EDC), data validation, database design, clinical trial protocols, regulatory compliance, and data quality management. Learners will also gain practical experience with industry-standard tools and real-world case studies, preparing them to handle complex data challenges in clinical trials.
By completing this course, you will receive a certification recognized by the industry, validating your skills and enhancing your career prospects. With flexible online learning, you can study at your own pace while gaining practical expertise that bridges the gap between academic knowledge and industry requirements.
Industry Aligned Curriculum
Covers all essential aspects of clinical data management, from data collection to database lock.
Industry Relevant Training
Learn the latest industry practices, guidelines, and software used in clinical trials.
Hands On Practical Exposure
Gain real-time experience with case studies, mock projects, and data handling exercises.
Flexible Online Learning
Access course content anytime, anywhere, at your own pace without disrupting your schedule.
Certification upon Completion
Earn a recognized certification to enhance your resume and career prospects.
Experienced Faculty Support
Learn from industry experts with years of practical clinical research and data management experience.
Career Focused Approach
Prepare for roles like Clinical Data Manager, Data Coordinator, or Clinical Research Associate with practical skills.
Clinical Data Management Course Syllabus [2025 Edition]
Clinical Data Management course content offered by ProRelix Education is structured to align with current regulatory frameworks and global clinical trial standards, with a strong focus on conceptual clarity, practical understanding, and job-oriented skills. The syllabus is designed to prepare learners for roles across clinical trials, data management, and pharmacovigilance.
1 : Introduction to Clinical Data & CDM
- What is clinical trial data
- Types and sources of clinical data
- Introduction to Clinical Data Management (CDM)
- Objectives & Importance of CDM in clinical trials
- Roles in Clinical Data Management
- Key Documents & guidelines related to CDM
2 : Overview of Clinical Data Management Workflow
- CDM lifecycle overview
- Key CDM activities
- Role of CDM across different trial stages
- CDM deliverables & process flow
3 : What is Protocol and Data Management Plan (DMP)
- What is a clinical trial protocol?
- Key protocol sections relevant to CDM
- Importance of protocol review in CDM
- What is Data Management Plan (DMP)?
- Objectives and components of DMP
4 : What is CRF and CRF Completion Guideline
- What is Case Report Form (CRF)?
- Importance of CRF in Clinical Research
- Paper CRF vs eCRF
- What is CRF Completion Guideline (CCG)?
- Importance of CCG for accurate data entry
5 : What is Clinical Data Management System (CDMS)
- Introduction to CDMS
- Role of CDMS in clinical trials & its types
- Key functionalities & benefits of CDMS
- Global library & edit check specifications
6 : Data Entry Process
- Introduction to paper CRF & e CRF data entry
- Data entry workflow
- Types of data entry
- Importance of Accurate Data Entry
- Data entry guidelines
7 : Introduction to Discrepancy Management
- What is a discrepancy
- Types of discrepancies
- Query generation and resolution
- Importance of discrepancy management
8 : Introduction to Medical Coding and Dictionaries
- What is medical coding?
- Importance of medical coding in clinical trials
- Introduction to coding dictionaries:
- MedDRA
- WHODrug
- Basic coding workflow
9 : Safety Database
- Introduction to safety database
- Components of safety database
- What is SAE reconciliation?
- SAE reconciliation process
10 : External Data
- Introduction to external data
- Types of external data
- Sources & importance of external data
11 : Manual Review and Quality Control (QC)
- What is manual data review?
- Importance of Data Quality in Clinical Trials
- Types of Manual Review
- Issue Communication & Resolution
12 : Manual Review and Quality Control (QC)
- What is database lock?
- Preconditions for database lock
- Steps involved in database lock
- Benefits of database lock
13 : Data Archival & Essential Terminologies
- Introduction & purpose of data archival
- Archival Media & Formats
- Types of Data to be Archived
- Archival storage Requirements
14 : Essential Terminologies
- Essential Clinical Data Management terminologies
What You Get After Clinical Research Course Done

On successful completion of the online Clinical Research course, you will receive a global-level certification from ProRelix Education. This certification strengthens your career path, enhances your professional credibility, and adds strong authority to your profile in the clinical research field. Backed by years of trust and expertise, this certification helps you unlock better career opportunities.
It reflects industry-relevant knowledge aligned with global clinical research standards. The certification supports career growth across CROs, pharmaceutical companies, and research organizations worldwide. It also demonstrates your commitment to continuous learning and professional excellence. Grab the opportunity!
Why This Courses Are Right For You
Clinical Research Focus
Focused content covering clinical trials, GCP, safety, and research workflows.
Regulatory-Aligned Learning
Courses structured around ICH-GCP principles and global compliance basics.
Concept-First Approach
Clear explanations that build strong fundamentals, not rote memorization.
Real-World Trial Scenarios
Practical scenarios explain how clinical studies run from start to close.
Trusted Since 2014
Over a decade of consistent clinical research education and credibility.
Built on 20+ Years Industry Expertise
Course content shaped by deep industry experience and applied knowledge.
Top Job Roles Opportunities After Completing a Clinical Data Management Course
1 : Clinical Data Coordinator
Supports data management activities by handling CRF review, data entry, query resolution, and coordination with clinical teams.
2 : Clinical Data Associate (CDA)
Focuses on data cleaning, discrepancy management, and maintaining high-quality clinical databases.
3 : Medical Coder
Assigns standardized medical codes (MedDRA, WHO-DD) to clinical trial data for analysis and regulatory submissions.
4 : Data Analyst
Analyzes clinical datasets to support statistical reporting, safety analysis, and decision-making.
5 : Database Programmer / Clinical Programmer
Designs and manages clinical databases using EDC systems and supports data extraction, validation checks, and reporting.
6 : EDC Specialist
Manages Electronic Data Capture systems, performs user setup, system testing, and supports study teams during trials.
7 : Quality Control (QC) Analyst – Clinical Data
Conducts data audits, reviews datasets, and ensures adherence to SOPs and regulatory guidelines.
8 : Clinical Data Entry Associate
An entry-level role focused on accurate data entry from source documents into EDC systems, basic data validation, and supporting data cleaning activities under supervision. This position is ideal for freshers starting a career in Clinical Data Management.
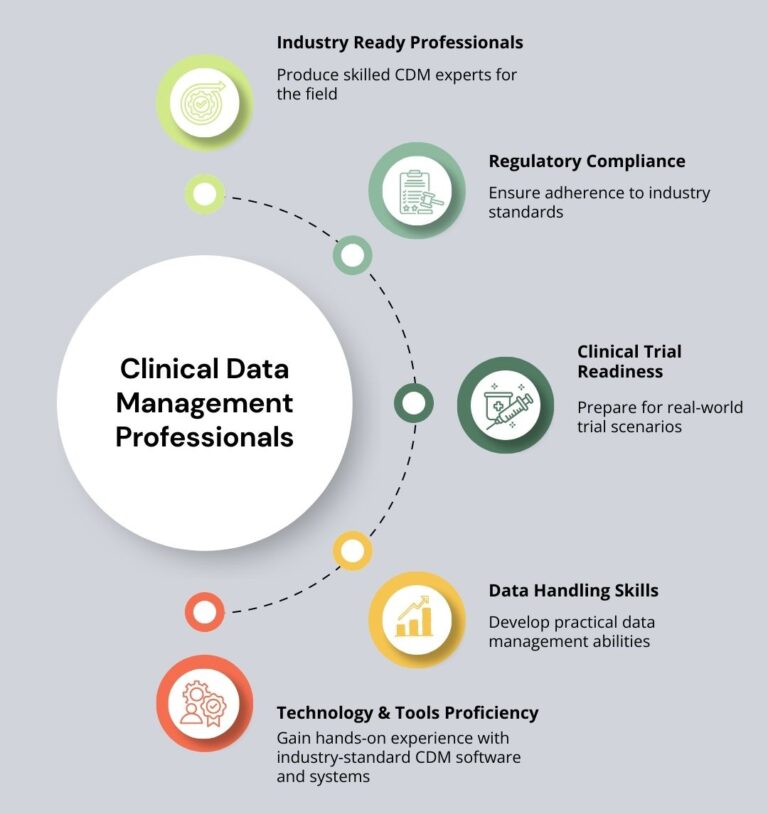
Industry-Ready Training Clinical Data Management Professionals
This program is designed to prepare aspiring and working professionals for real-world roles in Clinical Data Management (CDM). The training focuses on practical knowledge, hands-on experience, and industry-aligned processes to ensure learners are job-ready from day one.
The course covers core CDM concepts such as clinical trial data lifecycle, CRF design, data validation, discrepancy management, and database lock procedures, following global regulatory standards including ICH-GCP, FDA, and CDISC guidelines. Learners gain exposure to industry-used tools and workflows, helping them understand how data is managed across different phases of clinical trials.
With a strong emphasis on practical training, real-time case studies, and scenario-based learning, this program bridges the gap between theoretical knowledge and industry application. By the end of the training, participants develop the confidence, technical skills, and regulatory awareness required to succeed as Clinical Data Management professionals in CROs, pharmaceutical companies, and clinical research organizations.
Self Paced Online Clinical Research Course Offered by ProRelix Education
Advance Course in Clinical Research
ICH GCP : Good Clinical Practice
The Beginner's Course in Clinical Research
ICH GCP : Good Clinical Practice (Free)
The Beginner's Course in CDM
Frequently Ask Questions (FAQ's)
1. What is Clinical Data Management?
Clinical Data Management (CDM) involves organizing, validating, and ensuring the quality and integrity of clinical trial data throughout the study lifecycle.
2. Who can take this online CDM course?
Students from life sciences, pharmacy, biotechnology, nursing, and related backgrounds can enroll. Even career changers interested in clinical research can join.
3. Is the online course suitable for beginners?
Yes. Many online CDM courses are designed for beginners with no prior experience in clinical data management.
4. What skills will I learn in CDM course?
You’ll learn data collection and validation, query management, regulatory compliance principles, Electronic Data Capture (EDC), and data quality control.
5. Can I take CDM course fully online?
Yes. Most programs offer fully online learning options with flexible schedules.
6. Do I need any programming knowledge in clinical data management?
Basic data handling knowledge helps, but programming is not usually required for entry-level CDM courses.





