Awarded as “Best Clinical Research Institute in India”
Best Clinical Research Courses in India with Placement Assistant
ProRelix Education offers the Best Clinical Research Courses in India with placement assistant providing career opportunities at IQVIA, TCS, Cognizant & more. Covering CDM, Pharmacovigilance, ICH-GCP, clinical trials, Regulatory Affairs, medical writing, and drug development, the program builds industry-ready skills through expert-led training and global exposure.
Get Free Consultation
ProRelix Education is an award-winning institute offering Best Clinical Research Courses in India with placement opportunities with top-tier MNCs and leading CROs. The institute delivers both online and offline learning programs focused on hands-on practical training, real-world industry exposure, and adherence to global clinical research standards.
The training methodology is strengthened by recognized accreditations and strong industry collaborations. ProRelix Education signed an MoU with the Life Sciences Sector Skill Development Council (LSSSDC) in 2017 and is ISO 9001:2015 certified. The institute was honored as the “Best Institute for Clinical Research” at the Asia Education Summit & Awards and received a “Certificate of Commitment” from the Central Vigilance Commission of India in 2016. Additionally, ProRelix Education has signed agreements with multiple organizations across the UK, South Africa, the USA, and Peru to support global clinical research development initiatives, offering learners valuable international exposure.
With a strong emphasis on ethical research practices, job readiness, and continuous professional development, ProRelix Education empowers students and working professionals to build successful careers in the clinical research and healthcare industry.
Clinical Research Course Highlights
- Top Quality at the Best Price
- 100% Placement Assistance
- Unlimited Job Opportunity Calls
- Learn Directly from Industry Professionals
- Proven 90% Placement Success Rate
- Flexible Payment Plans Available
- 10+ Years of Industry Experience
- Own ProRelix Research CRO for Internship Opportunities
- Development in Interview, CV, and Presentation Skills
What Is Clinical Research and Why It Matters in Modern Medicine
Clinical research is a specialized field of healthcare focused on evaluating new drugs and treatment approaches through carefully designed trials. These studies are conducted on both healthy volunteers and patients in the early stages to assess safety, effectiveness, and appropriate therapeutic use. The primary goal of clinical research is to establish effective treatment strategies for serious and life-threatening diseases. Such research is carried out at accredited medical and research centers, enabling healthcare professionals to enhance patient outcomes through innovative therapies.
The scope of a clinical research career is rapidly expanding in India and globally, driven by rising clinical trials, pharmaceutical innovation, and regulatory compliance needs. Professionals can build rewarding careers across pharmaceuticals, biotechnology, Contract Research Organizations( CROs), medical devices, pharmacovigilance, data management, and regulatory affairs, with opportunities for global exposure, career stability, and continuous growth. With healthcare becoming increasingly research-driven, careers in the clinical research sector offer long-term relevance, impact, and professional advancement.
Offline Fulltime Clinical Research Course in India with Placement Assistant
PGDCR- Post Graduate Diploma In Clinical Research (Full Time)
Modes of Teaching
Offline Classroom
Course Duration
04 Months
Courses Fees
40K -60K
Placement
100% Assistance
Student Trained
10000+
Online Fulltime Clinical Research Course in India with Placement Assistant
Modes of Teaching
Online Live
Course Duration
04 Months
Courses Fees
40K -60K
Placement
100% Assistance
Student Trained
10000+
ProRelix Education offers a full-time PG Diploma in Clinical Research Course in Pune, featuring affordable fees and placement opportunities with leading companies such as IQVIA, Lybra Clinical Research, Tata Consultancy Services, Wellbeing Research Centre, Raptim Research, ProRelix Research, Parexel, IKS Health, Sahyadri Hospitals Cognizant and more. This program, also known as the PGDCR: Post Graduate Diploma In Clinical Research, has a duration of 6 months and is available through both online live classes and offline classroom sessions in Karvenagar, Pune with placement opportunitis. The course fees range from 40,000 to 50,000 INR, depending on the selected modules and internship program. Many students have benefited from this course and its internship opportunities, securing placements in top companies.
PGDCR Clinical Research Course Syllabus [2025 Edition]
PGDCR Clinical research course content offered by ProRelix Education is structured to align with current regulatory frameworks and global clinical trial standards, with a strong focus on conceptual clarity, practical understanding, and job-oriented skills. The syllabus is designed to prepare learners for roles across clinical trials, data management, and pharmacovigilance.
1 : Fundamentals of Clinical Research
- Introduction to Clinical Research
- History & evolution of Clinical Research
- Drug development process and preclinical studie
- Phases of clinical trials and BA/BE studies
- Stakeholders in clinical research and their roles (Sponsor, Investigator, CRO, Ethics committees etc.
- Types of clinical trials and basic trial designs
- Regulatory framework and clinical trial registries
- Ethics in clinical research and informed consent
- Essential documents (Protocol, ICF, CRF, IB, etc.)
- ICH GCP E6 R
- Good Documentation Practices and ALCOA
- Basics of safety reporting
- Terminologies related to Clinical Research
2 : Clinical Research Operations
- Site selection & feasibility
- Initiation and ethics approvals
- Subject recruitment and clinical trial conduct
- Monitoring activities and source data verificatio
- Types of monitoring
- Investigational product management and accountabilit
- Protocol deviations and safety reporting
- Quality management, audits, and inspections
- Trial close-out, documentation, and archiving
3 : Clinical Data Management
- Introduction to Clinical Data Management & its role in clinical trials
- Clinical data management process flow and data lifecycle
- Data collection methods and instruments
- Data management team roles and responsibilities
- Data Management Plan (DMP)
- CRF/eCRF design principles and CDASH standards
- Edit check specifications and validation rules
- EDC systems, database build, and User Acceptance Testing (UAT)
- Data entry, validation, and query management
- Medical coding and safety data reconciliation
- Computer System Validation (CSV) and 21 CFR Part 11
- Database lock, data transfer, and study close-out activities
- CDM quality management, documentation, and compliance
4 : Pharmacovigilance
- Introduction to Pharmacovigilance
- Adverse events, special situations, and case validity
- Causality & seriousness assessment
- Individual Case Safety Reports (ICSRs)
- Case Processing and Coding
- Safety Reporting & timelines
- Signal Detection and Risk Management
- Pharmacovigilance Systems and Quality Management
- Pharmacovigilance regulations in India and global context
- Pharmacovigilance Databases and Tools
- Common terminologies related to pharmacovigilance
5 : Regulatory Affairs
- Introduction to Pharmacovigilance
- Adverse events, special situations, and case validity
- Causality & seriousness assessment
- Individual Case Safety Reports (ICSRs)
- Case Processing and Coding
- Safety Reporting & timelines
- Signal Detection and Risk Management
- Pharmacovigilance Systems and Quality Management
- Pharmacovigilance regulations in India and global context
- Pharmacovigilance Databases and Tools
- Common terminologies related to pharmacovigilance
Our Placed Students
Our students have placements with leading companies worldwide.









Our Placement Companies













PGDCR Offline/Online Clinical Research Courses Duration & Eligibility
Understanding the duration and eligibility of Clinical Research Courses is essential for learners aiming to build a strong foundation in this high-growth domain. These programs are structured to blend theoretical knowledge with practical exposure, preparing candidates for global clinical research roles.
Course Duration
Course Eligibility
- Post Graduate Diploma in Clinical Research (Full-Time): 6 months
- 4 months of structured training
- 2 months of internship with hands-on, industry-level experience
- Graduates in Life Sciences
- Pharmacy graduates
- Biotechnology graduates
- Candidates from allied healthcare & related scientific backgrounds
PGDCR Clinical Research Course Fees
Clinical research courses play a vital role in building industry-ready knowledge and practical expertise for a successful life-sciences career. To remove financial barriers, ProRelix Education offers transparent and affordable Clinical Research Courses fees, along with flexible two-installment payment options, making quality education accessible while supporting confident career growth.
Course Name | Mode of Training | Course Type | Fees Range |
PGDCR – Post Graduate Diploma in Clinical Research | Offline (Classroom) | Full Time | ₹40,000 – ₹60,000 |
PGDCR – Post Graduate Diploma in Clinical Research | Online(Live) | Full Time | ₹40,000 – ₹60,000 |
What Our Students Say
Read student reviews about their learning journey at ProRelix Education
Clinical Research Training: Learn from Industry Leaders
ProRelix Education offers advanced clinical research training led by industry experts and highly supportive trainers. With limited seats, we ensure personalized attention, allowing each student to gain hands-on experience & mastery of essential skills. Our structured learning pathways, real-world project exposure, & placement-focused guidance empower aspiring professionals to excel in global clinical trials & build successful careers in the clinical research industry.
Clinical Research Course Certification
🏅 Get Certified by ProRelix Education
Successfully completing the Clinical Research Course at ProRelix Education awards you a globally recognized certification, reflecting your advanced expertise and readiness to excel in the clinical research industry.
- Validates your in-depth knowledge and practical skills in clinical research
- Globally recognized and valued by leading employers and international industry partners
- Strengthens your resume and opens doors to advanced career opportunities
- Demonstrates your commitment to professional growth and industry excellence

Clinical Research Salaries in India : Career Insights
The clinical research field provides competitive salary prospects for professionals at all stages of their careers. Fresh entrants, such as clinical research coordinators or junior CRAs, typically earn between Rs 1.8LPA and Rs 4LPA. With experience, mid-level roles like senior CRAs, project managers, and clinical trial associates can command salaries ranging from Rs 6LPA to Rs 10LPA. Senior leadership positions, including directors of clinical operations or vice presidents of clinical research, often see compensation exceeding Rs 12LPA. Actual salaries may differ based on factors such as industry type, location, organizational scale, and academic qualifications.
Designation | Tentative Annual Salary (INR) |
Clinical Research Associate (CRA) | ₹3 – 12 LPA |
Clinical Research Coordinator (CRC) | ₹2.5 – 8 LPA |
Clinical Project Manager | ₹12 – 25 LPA |
Regulatory Affairs Specialist | ₹3 – 15 LPA |
Data Manager / Clinical Data Analyst | ₹4 – 16 LPA |
Clinical Trial Assistant (CTA) | ₹2.5 – 4.5 LPA |
Pharmacovigilance / Drug Safety Officer | ₹3.5 – 14 LPA |
Medical Writer | ₹4 – 18 LPA |
Quality Assurance (QA) Specialist | ₹3.5 – 15 LPA |
Biostatistician | ₹6 – 30 LPA |
Quality Assurance Associate | ₹3 – 5 LPA |
Regulatory Associate | ₹3 – 5.5 LPA |
Investigator (Clinical Trials) | ₹15 – 40 LPA+ |
Business Development Executive (Clinical Research) | ₹4 – 20 LPA |
Trainer (Clinical Research / PV / CDM) | ₹6 – 15 LPA |
Career In Clinical Research: Opportunities and Growth
A Career In Clinical Research offers a promising path for professionals passionate about advancing healthcare and life sciences. It provides exposure to clinical trials, regulatory compliance, and medical innovations while offering strong growth potential & attractive salary prospects. Professionals gain hands-on experience in clinical operations, data management,& patient safety, making them highly valuable in pharmaceutical companies, contract research organizations (CROs), & research institutions. With increasing demand for new therapies & medical devices, this field offers a stable & rewarding career trajectory.
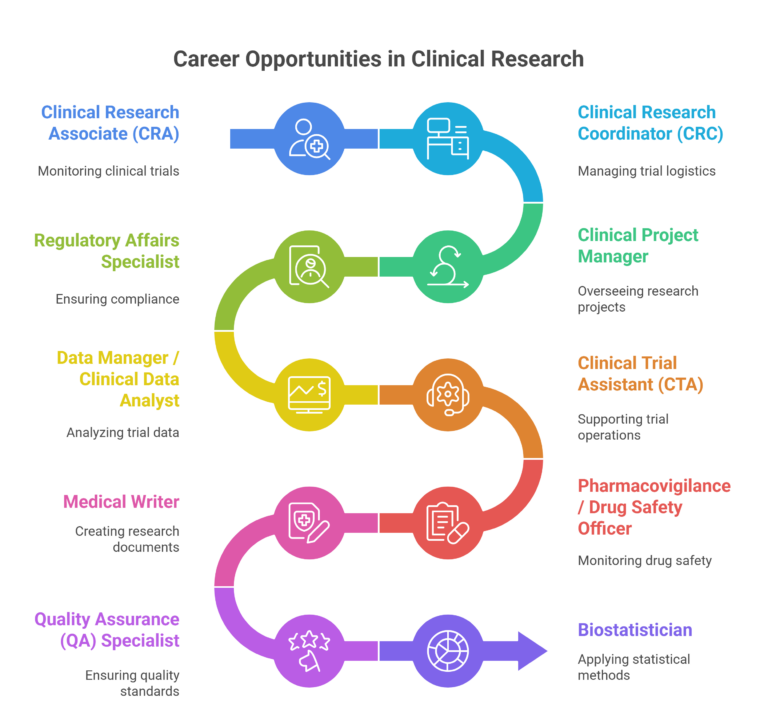
Start a Promising Clinical Research Career in Key Sectors
Embarking on a career in clinical research opens doors to a dynamic and rewarding profession, offering opportunities to contribute to the development of life-saving therapies and innovative healthcare solutions. With a growing demand for skilled professionals, a career in clinical research promises not only professional growth but also the chance to make a meaningful impact on global health.Listed below are the sectors you can work in after completing a PG Diploma in Clinical Research:
- Pharmaceutical Industry
- Biotechnology
- Medical Devices
- Healthcare Institutions
- Contract Research Organizations (CROs)
- Regulatory and Quality Assurance
- Pharmacovigilance Centers
- IT Companies in Healthcare
- Central Laboratories
- Academic & Research Institutes
Why Choose ProRelix Education for Best Clinical Research Career
Industry Focused Curriculum
We Offers complete courses aligned with global clinical research standards.
Expert Faculty
Learn from seasoned professionals with real-world clinical trial experience.
Hands On Training
Practical exposure to clinical research processes and documentation.
Ethical & Regulatory Guidance
Strong emphasis on ethics and compliance in clinical trials.
Career Oriented Approach
Programs designed to enhance employability in top clinical research organizations.
Own ProRelix Research CRO
Hands-On Internship Opportunities with ProRelix Research, Our Own CRO
Career Advancement
The Growing Scope and Impact of Clinical Research
India has emerged as a global hub for clinical trials, leading many multinational companies to establish research and development centers in the country. This growth is driven by the vital role of clinical trials in evaluating the safety and effectiveness of medicines, medical devices, and treatment options before they are introduced to the market. As a result, the demand for skilled and trained professionals has increased significantly, creating strong career opportunities in clinical research. This demand is not limited to India alone but extends worldwide, with excellent job prospects in regions such as the USA, Europe, and Singapore. With over 2.5 lakh global job openings and the clinical trials market expected to reach USD 68.9 billion by 2026 at a CAGR of 5.7%, the scope of clinical research is set to expand rapidly in the coming years.

Frequently Ask Questions (FAQ's)
What clinical research courses are available in India?
Clinical research courses available in India include certificate programs, diploma courses, and postgraduate diplomas in Clinical Research, Clinical Data Management, Pharmacovigilance, Regulatory Affairs, Medical Writing, and ICH-GCP, offered through online, offline, and blended learning modes.
Who can enroll in a Clinical Research Course?
Life science, pharmacy, medical, biotechnology graduates, and working professionals are eligible to enroll.
What are the career options after a Clinical Research Course?
Career options include Clinical Research Associate, Clinical Trial Coordinator, Data Manager, and Regulatory Associate.
Is a Clinical Research Course job-oriented?
Yes, the course focuses on practical skills, real-world projects, and industry-ready training.
Does the Clinical Research Course include practical training?
Many institutes, including ProRelix Education, provide placement assistance to support career entry.
Which industries hire clinical research professionals?
Pharmaceutical companies, CROs, biotech firms, medical device companies, and healthcare organizations hire them.




